 Gastrointestinal side effects tend to be more problematic in the first few months of therapy. He was treated with blood and platelet transfusions and discharged eight days later with modest improvement in peripheral blood count. Ten days later he was admitted to the hospital with malaise, lethargy, and deterioration in blood count. 
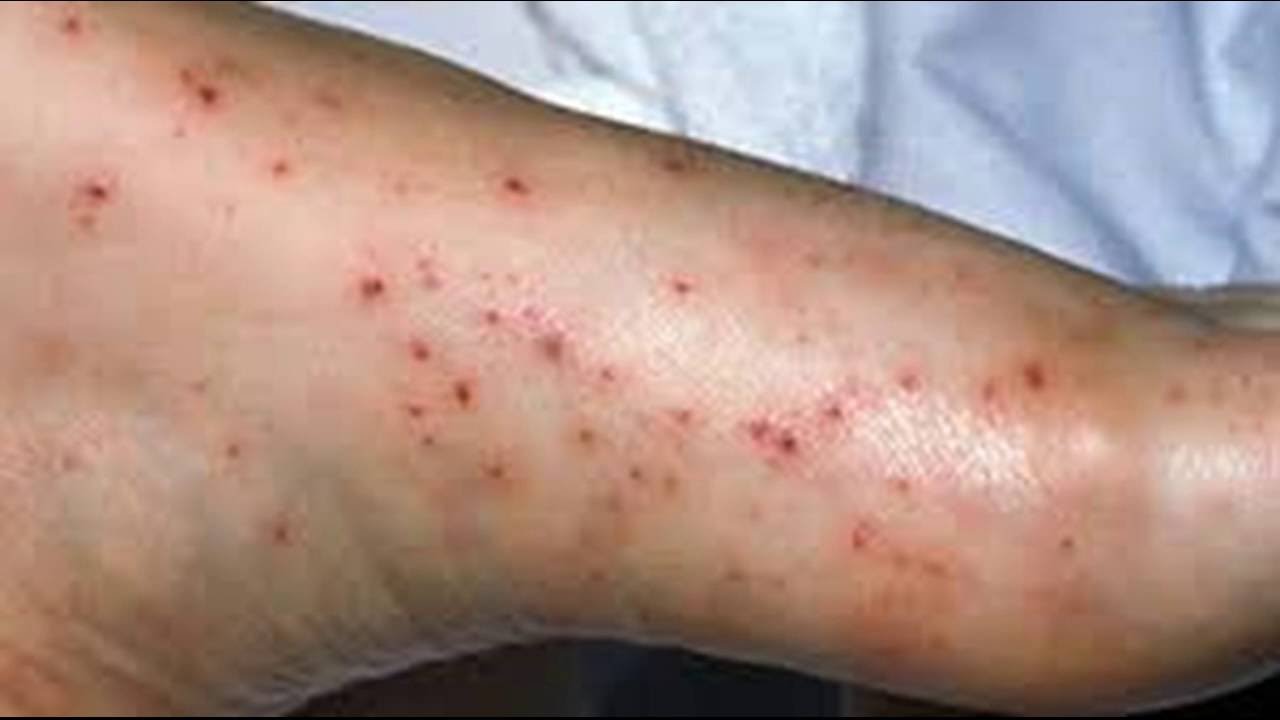
Ten weeks after starting azathioprine 100 mg per day, a full blood count (during routine monitoring) showed moderate pancytopenia. Bone marrow aplasia has been reported in patients with normal TPMT activity.Ī 55-year-old male with pompholyx and deficiency of erythrocyte thiopurine methyltransferase experienced pancytopenia coincident with azathioprine therapy. Death associated with pancytopenia has been reported in patients with absent TPMT activity receiving azathioprine. In patients with low TPMT activity, there is an increase in 6-thioguanine nucleotide (6-TGN) concentrations, a cytotoxic metabolite which suppresses purine synthesis. There are data to support an increased risk of bone marrow aplasia in patients with very low or absent thiopurine methyltransferase (TPMT) activity. Leukopenia (less than 2500/mm3 ) has been reported in 16% of renal homograft recipients and 5.3% of rheumatoid arthritis patients. Leukopenia (any degree) has been reported in greater than 50% of renal homograft recipients and 28% of rheumatoid arthritis patients. Rare (Less than 0.1%): Agranulocytosis, aplastic anemia, erythroid hypoplasia, megaloblastic anemia, pancytopeniaįrequency not reported: Bleeding, increased mean corpuscular volume and red cell hemoglobin content (reversible, dose-related) īone marrow suppression is dose related and is the most common cause for dosage reductions. Very common (10% or more): Depression of bone marrow function, leukopeniaĬommon (1% to 10%): Anemia, thrombocytopenia Hematologic toxicities and neoplasia were reported more often in renal homograft recipients than in patients using azathioprine for rheumatoid arthritis. Frequency and severity of side effects depend on dose and duration of azathioprine as well as on underlying disease or concomitant therapies. Risk of secondary infection and malignancy is also significant. The principal and potentially serious toxic effects of azathioprine (the active ingredient contained in Azasan) are hematologic and gastrointestinal. Also, your health care professional may be able to tell you about ways to prevent or reduce some of these side effects.Ĭheck with your health care professional if any of the following side effects continue or are bothersome or if you have any questions about them: More commonĪpplies to azathioprine: compounding powder, intravenous powder for injection, oral tablet. These side effects may go away during treatment as your body adjusts to the medicine. Some side effects of azathioprine may occur that usually do not need medical attention.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
